This is a preview version of the report. The first two chapters are available for you to browse. To view the full version, click the button below to purchase:
INTRODUCTION
Cancer will affect half the global population by 2050. By then it will be by far the commonest cause of death. On World Cancer Day 2022, the UK Health Minister, Sajid Javid, announced an ambitious ten-year plan to elevate to UK’s cancer care system to ‘the best in Europe’. Borrowing from U.S. President Richard Nixon’s ‘War on Cancer’ over half a century earlier, the UK Government pledged a range of priorities, from improving early diagnosis to boosting research on mRNA vaccines and re-doubling efforts to increase the cancer workforce.
Despite the bold platitudes, cancer remains one of the UK’s greatest public health challenges. In 2023, cancer waiting times in England were the worst since records began, with the Less Survivable Cancers Taskforce finding that the UK is near the bottom of the pack on cancer survival rates in the developed world. Where the UK lags in diagnosis, it fails to compensate in treatment. A recent report found that innovative cancer drugs face major delays in getting to patients and proton beam therapy is severely rationed with no plans for expansion.
While global cancer incidence is expected to nearly double to 35 million cases worldwide by 2050, this trend will intersect with a disease management landscape that is rapidly evolving. Owing to recent developments in computing power, artificial intelligence has increasingly been deployed in clinical settings, accelerating advancements in drug discovery and development, diagnosis, treatment, and patient management. In this highly dynamic and uncertain environment, the horizon scanning necessary to make effective long-term decisions for the future of cancer care can prove particularly challenging.
Vision 2050 emerges as a groundbreaking document to help bridge this gap, providing a peek over the horizon with the hope of better equipping policymakers, biomedical innovators and healthcare managers to make decisions for the future of cancer care. This report leverages the collective intelligence of leading AI systems—ChatGPT 4, Google Gemini, and Claude.ai—enhanced by the expertise of cancer specialist Karol Sikora. Together, they offer a vision of cancer care's evolution by 2050. With insights refined by AI and human expertise, the report not only forecasts technological advancements but also anticipates shifts in patient care paradigms, focusing on accessibility, equality, and cultural sensitivity. This introduction serves as a gateway to a document that embodies a multidisciplinary endeavour to envision a future where cancer care is more effective, personalized, and inclusive, guided by the latest in AI and medical insights.
Danny Al-Khafaji, CEO, Future Science
I’ve specialised in cancer care for nearly 50 years. From my days as a young registrar at St Batholomew’s Hospital in London, I’ve worked in a diverse range of areas of cancer. I have been involved in research, published over 500 papers, written 20 books on the subject and edited Britain’s standard postgraduate textbook on the treatment of cancer for 35 years. I’ve been a consultant oncologist in the NHS for 45 years, been clinical director of a major cancer centre in West London and treated countless of patients. I was the creator and founding dean of the University of Buckingham Medical School. I’ve been an adviser to big pharmaceutical companies, been on the board of major cancer charities and built several cancer centres in the UK and around the world. I’ve also witnessed first-hand the plight of cancer patients in some of the poorest countries of Sub-Saharan Africa when working as Director of the WHO Cancer Programme.
I’ve always viewed AI with great suspicion. But it has arrived and is here to stay. This is my first interaction with it. I am truly amazed at its power - beyond anything I imagined. We all know there is a severe ability gradient with age for computers, mobile phones, and AI use. My eleven-year-old grand-daughter is far faster and wiser in using them than an aging physician like me. This experiment arose after much discussion with Danny Al-Khafaji and James Drake at the Future Medicine group. 20 years ago, I organised a meeting of cancer experts at a delightful riverside hotel just outside Henley-on-Thames to predict the future of cancer care. It was published in 2004 as a special issue of their journal.
This offering is the same concept but with a major difference. The experts are not human but three AI engines. No pleasant drinks by the river, heated discussions over delicious meals or philosophical, futuristic conversations after midnight took place. I have asked fifty questions to the three large language models that dominate the AI space. Here are their answers, lightly edited by myself, simply to avoid their repetition and rather staccato dialogue. No single engine was best for all the answers it gave. All three were scrupulously polite and I thought extremely honest about their limitations at predicting the future. They also surprised me by their concern with inequalities in access to healthcare.
Futurology is just fascinating. The trouble for both human and AI experts is that we all see more of the same – a sort of continuum of progress. But the future is simply not like that – there will be totally unpredictable step changes in progress. No computer could have foreseen the development of CT scanning in 1972, the emergence of the now familiar PCR test for tiny specific DNA fragments in 1986 or the total sequencing of the human genome in 2003. The same applies to the vast array of specific tyrosine kinase inhibitors and monoclonal antibodies we now use routinely for cancer. But here we have a synthesis of AI wisdom giving us a vision of cancer care in 2050.
I feel like the little old man behind the curtain in the 1939 classic film The Wizard of Oz. I saw it when I was six years old and remember vividly being frightened out of my skin by the Wizard himself - a fierce looking but rather primitive computer who blared out the correct answer to any question. Dorothy played by Judy Garland, dares to challenge the Wizard:
Dorothy: Who are you?
The Wizard of Oz: Who, ah, ah... I am the Great and Powerful... Wizard, of Oz.
Dorothy: YOU are? I don't believe you!
My fear dissolved when she drew back the curtains and revealed a little old man shouting the answers down a megaphone, but I still get nightmares about the Wicked Witch of the East. This is different – I am not the author of the answers – simply the translator and editor. I‘m the old man with the megaphone but shouting the computer’s answers.
We both would be most interested on your feedback. We plan to produce bespoke horizon scanning using the same methodology to drill down the future of all the components that will bring change to how we prevent, diagnose and treat and care for cancer patients.
Karol Sikora, consultant oncologist
NUMBERS
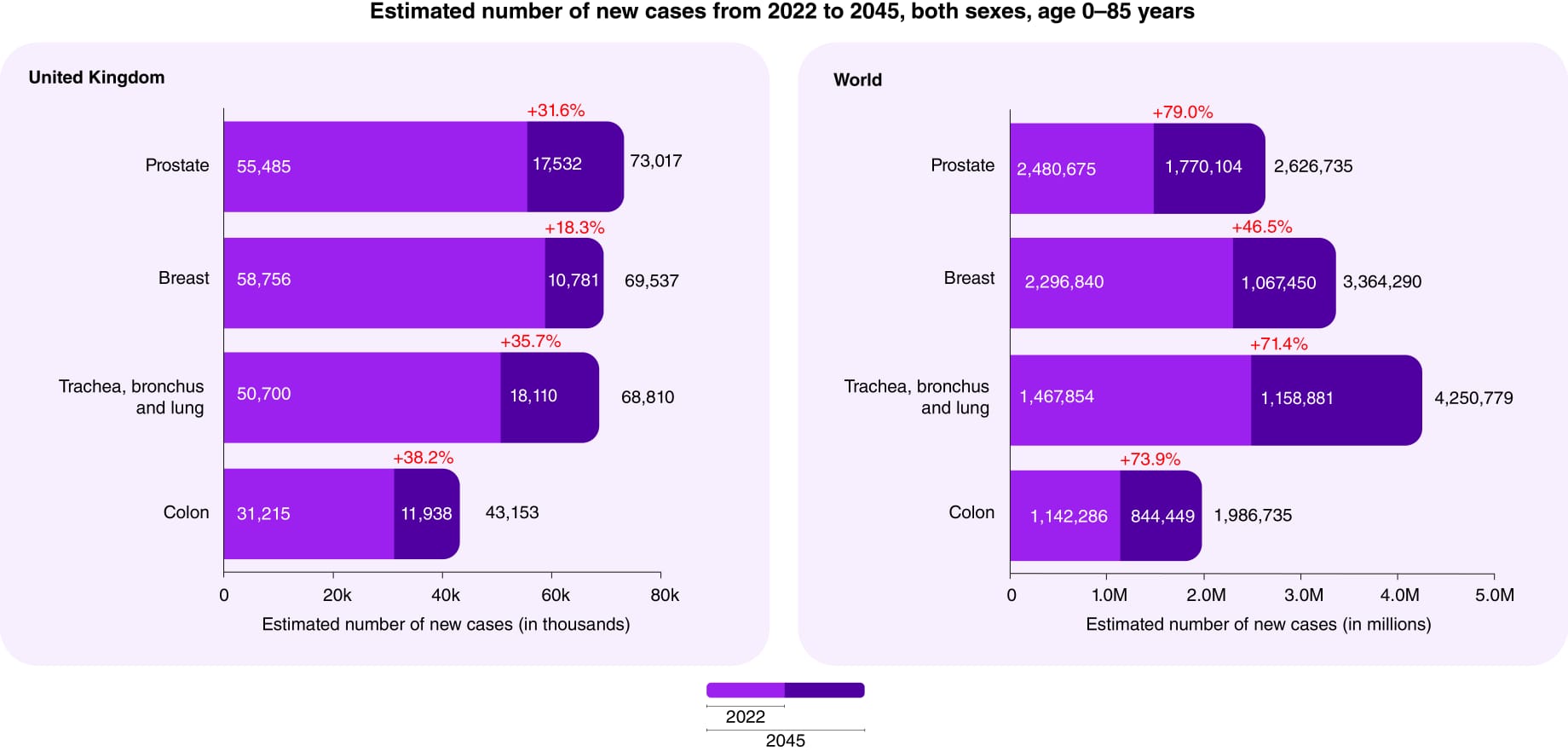
Globally, the number of new cancer patients each year is expected to rise from 18m in 2023 in a population of 8bn people to 35m by 2050 in a population of 10bn people – a 77% increase. This is primarily due to the aging of the population since the risk of cancer increases considerably with age. Also, as life expectancy increases globally, more people will live long enough to develop cancer. The increase in cancer incidence may be more pronounced in developing countries. This is partly due to these countries undergoing rapid societal and economic changes, leading to lifestyle patterns (such as diet, physical activity, and tobacco use) that are associated with higher cancer risk. Additionally, these regions are experiencing improvements in healthcare infrastructure and access, leading to better detection and reporting. Even if dramatic lifestyle changes occurred immediately, it would take many years for any change to be reflected in the overall cancer burden. The numbers are simply locked into our future, certainly over a 25-year span.
Impact of lifestyle and environmental factors:
Changes in lifestyle, such as increased consumption of processed foods, physical exercise, alcohol use and obesity profoundly affect the epidemiology of cancer. Unfortunately, there is not enough information to predict specific annual changes in the number of cancer patients by world region. Some general points:
Growth rates of cancer incidence will likely differ significantly across regions based on population aging, adoption of unhealthy lifestyles, early detection capacities and case notification. Developed regions may see slower increases in annual cases with population stabilization, but with a higher cost of treatment per case. Lower-middle income regions could see rapid rises in new cases given growing and aging populations.
While a global rise of 77% in new cancer cases by 2050 is widely predicted, the specific annual changes for different world regions are more nuanced and depend on various factors. Here's a breakdown of what we know:
Overall Trends
- Africa: The fastest-growing cancer burden, potentially more than doubling by 2050 due to increasing population and urbanization.
- Asia: Expected to face the highest absolute number of new cases due to its large population, although the annual increase might be lower than Africa.
- Latin America and the Caribbean: A projected moderate increase, influenced by aging populations and lifestyle changes.
- Europe and North America: Slower increase due to already advanced healthcare systems and stabilizing populations, though still significant due to aging populations.
- Oceania: The smallest increase due to a low and slow-growing population, but still facing challenges due to existing disparities in access to healthcare.
Regional variation: The annual changes will vary based on the types of cancer prevalent in each region. For example, lung cancer might show a higher increase in areas with high smoking rates, while physical inactivity, and the ongoing high rates of smoking in certain regions, can lead to an increase in certain types of cancers. Environmental factors, including pollution and exposure to carcinogens, also play a smaller but significant role.
In regions with robust cancer screening and prevention programs, there might be an initial increase in reported cancer cases due to improved detection. Over time, however, these programmes can lead to a decrease in certain types of cancers, as seen with cervical cancer and the impact of HPV vaccination. The types of cancers that are most prevalent can vary by region due to genetic, environmental, and lifestyle factors. For instance, gastric cancer is more common in East Asia, while colorectal cancer rates are much higher in Western countries.
The COVID-19 pandemic has led to delays in diagnosis that cannot reliably be predicted in different healthcare systems. Income level, healthcare access, and lifestyle choices significantly impact cancer incidence. Low-middle income countries will face steeper increases due to limited awareness, prevention resources, and treatment options. Regions with a rapidly aging population, like Europe and parts of Asia, will likely see a bigger rise in incidence due to the increased risk associated with age.
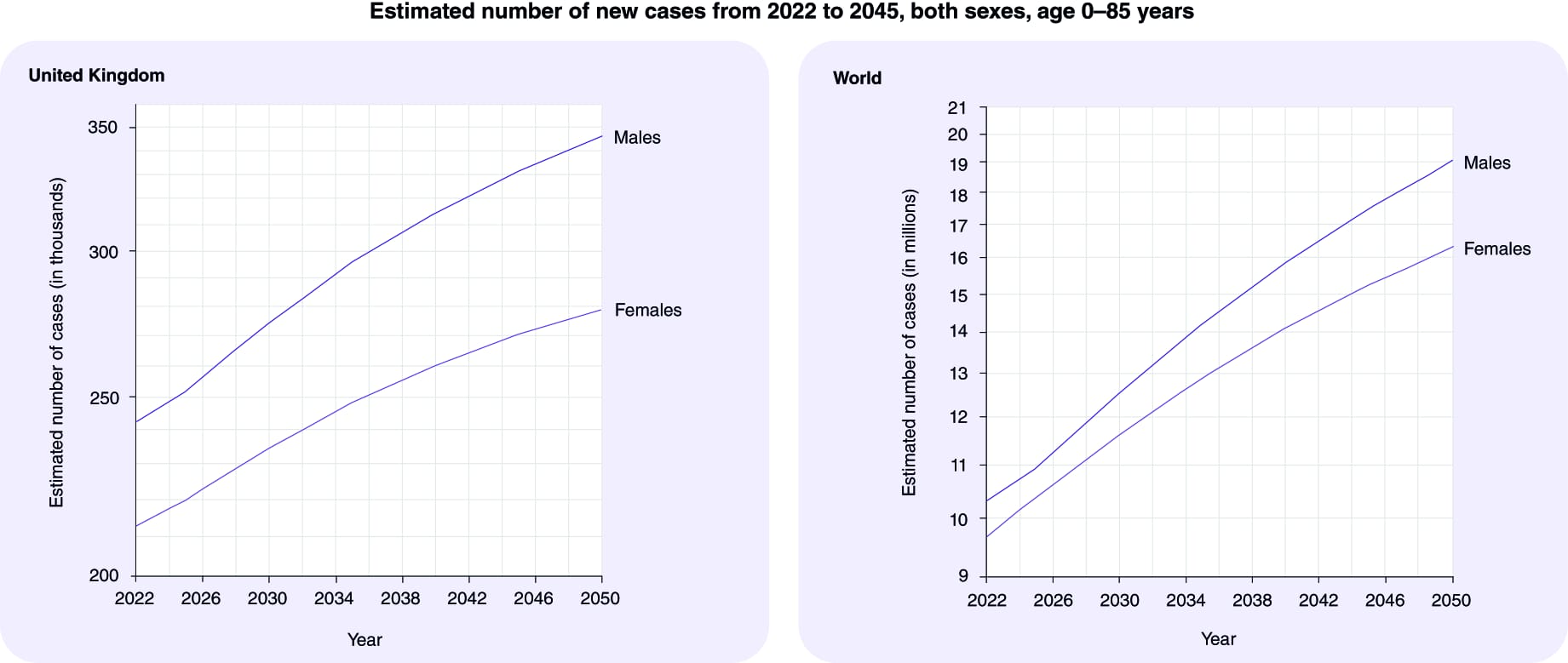
- Which countries currently have a higher and lower than average cancer incidence for the four common types and how will this change?
Breast Cancer
- Higher Incidence: Developed countries, particularly in North America and Western Europe, tend to have higher rates of breast cancer and this will continue. This is often attributed to lifestyle factors, reproductive behaviours (having fewer children and having them later in life), and widespread screening programs that lead to early detection.
- Lower Incidence: African and Asian countries generally have lower rates of breast cancer. However, these rates are increasing as these countries undergo economic development and adopt Western lifestyle patterns. Making sound predictions would require granular data on risk factors, demographics, detection rates, and health systems for each major country over the target time-frame.
Lung cancer
- Higher incidence: high income countries like US, Europe, Japan and East Asia with historically high smoking rates, and in some areas, air pollution.
- Lower Incidence: Sub-Saharan Africa and parts of South America have lower lung cancer rates, which can be correlated with lower smoking rates and other environmental factors. The incidence of lung cancer will decline in countries with effective tobacco control such as North America and much of Europe.
Prostate cancer
- Higher Incidence: North America, the Caribbean, and Northern Europe see higher rates of prostate cancer, partly due to widespread screening practices using serum prostate specific antigen (PSA) that lead to early detection.
- Lower Incidence: Asian countries generally have lower rates of prostate cancer.: Global trends in prostate cancer incidence will likely be influenced by changes in screening practices and the aging population. By 2050, these trends may shift due to various factors including screening tests with greater sensitivity and specificity.
Colon cancer
- Higher Incidence: North America, Europe, and Australia have higher rates of colorectal cancer, which are often linked to dietary factors (high consumption of red and processed meat, low fibre intake), obesity, and sedentary lifestyles.
- Lower Incidence: African and South Asian countries have lower rates, though these rates are rising with the adoption of Western diets and lifestyle changes.
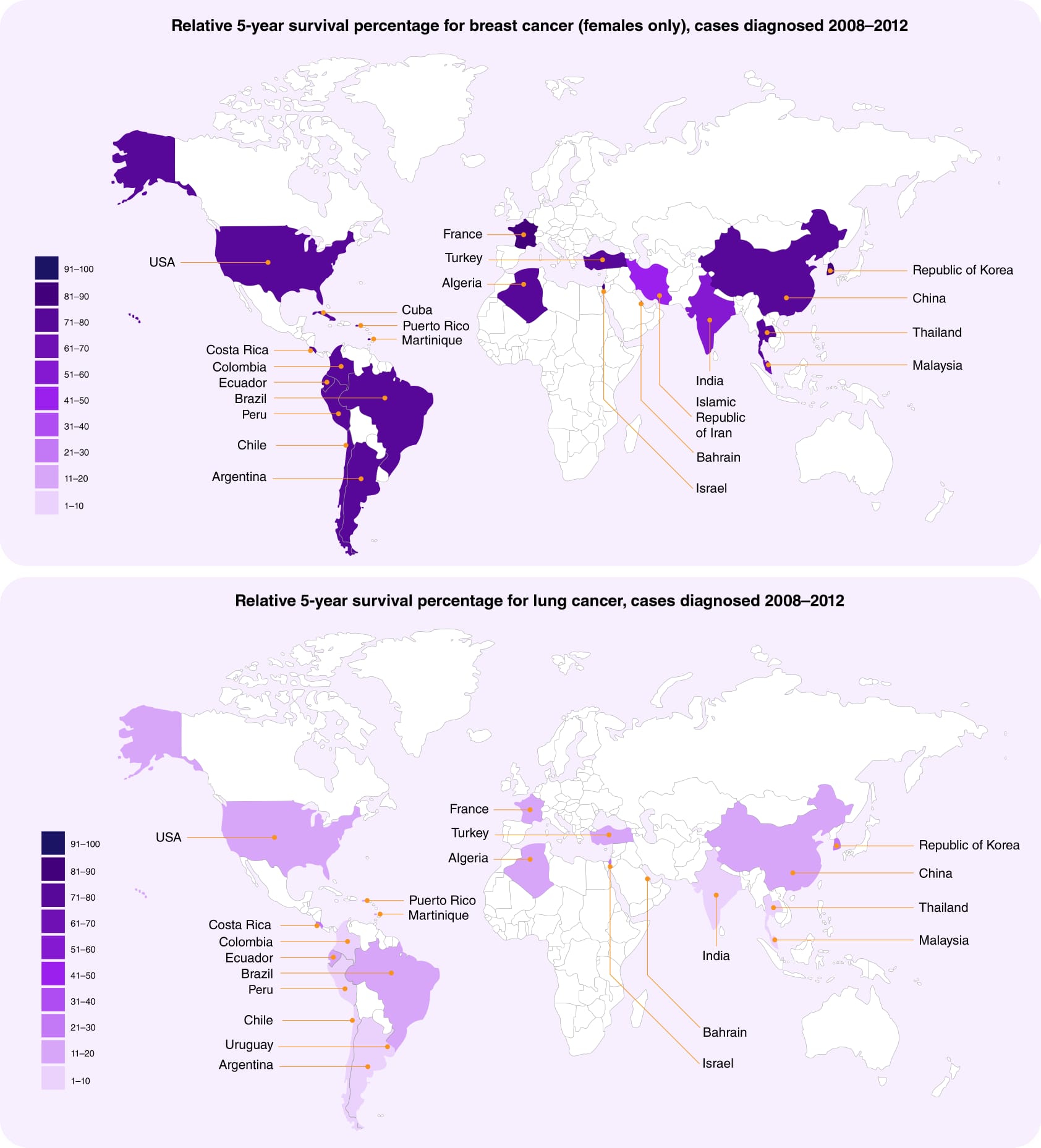
- Which of the OECD countries have the best outcomes for common cancers?
When evaluating "best outcomes," it's important to consider several indicators, including survival rates, mortality rates, and the quality-of-life post-treatment as well as life years lost. Here's an overview of OECD (Organisation for economic co-operation and development) countries generally recognized for their good cancer outcomes for the four most common cancers (breast, lung, colorectal, and prostate):
Based on data from the OECD, a few countries stand out as having some of the best outcomes for common cancers. Iceland, Japan, and South Korea tend to have very high 5-year relative cancer survival rates across all four major cancer types. Determining the "best" outcomes for common cancers across OECD countries requires considering multiple factors, as there's no single metric. However, based on several key indicators, some countries generally perform well:
High 5-year Survival Rates:
- Denmark: achieves top rank for breast, colorectal, and prostate cancer survival.
- Japan: excels in stomach and oesophageal cancer survival which can be attributed to early detection programs, advanced medical technology, and a healthcare system that provides universal coverage.
- South Korea: has made significant strides in cancer outcomes, especially in stomach and liver cancers. This is partly due to widespread screening programmes and advances in medical technology.
- Australia and New Zealand: Both countries show strong outcomes in cancer treatment, particularly for melanoma and breast cancer. These outcomes are supported by comprehensive cancer control programs and high-quality healthcare systems.
- Scandinavian Countries (Norway, Sweden, Denmark, Finland): These countries are known for their efficient healthcare systems and high cancer survival rates, particularly for prostate and breast cancer. Sweden prioritizes patient autonomy and shared decision-making in cancer care delivery.
- Canada and the United States: These countries typically have high survival rates for various cancers, indicate the percentage of patients who are still alive 5 years after their diagnosis compared to the general population. Their rates range from around 80-95% for some cancers. Interestingly, the United States lags despite highest spending - an example of how greater health expenditures don't always correlate with public health performance
- Switzerland also performs very well, with 5-year survival rates consistently above the OECD average. For example, Switzerland's survival is 87% for breast cancer and 66% for lung cancer. In general, the European countries with strong universal healthcare systems tend to have the most impressive outcomes.
- Netherlands: demonstrates active colorectal and cervical cancer screening initiatives.
- Germany: offers a sophisticated healthcare system with high-quality cancer treatments.
- UK: performs surprisingly badly in terms of survival despite having excellent universal coverage. The reason for this seems to be significant delays caused by poor system access and under capacity of diagnostic facilities and not the quality of actual treatment.
It's important to note that performance may vary by specific cancer type and individual circumstances. Additionally, factors like healthcare costs, sociodemographic inequalities, and geographic disparities can influence outcomes within a country. Considering all these factors, it's difficult to pinpoint a single "best" performer. However, the identified countries generally demonstrate a combination of successful prevention, early detection, advanced treatment access, and comprehensive care, leading to positive cancer outcomes for their populations.
- How will novel cancer preventive strategies impact on the overall epidemiology?
Novel cancer preventive strategies are poised to significantly impact the overall epidemiology of cancer in several ways. By reducing the incidence of new cancer cases and potentially lowering cancer mortality rates, these strategies can lead to profound shifts in cancer trends globally. Here are key areas where these impacts may be most evident:
Reduction in cancer incidence:
- Advances in genetic testing and personalized risk assessments can lead to targeted prevention strategies for individuals at high risk.
- Improved lifestyle interventions, including diet, exercise, and smoking cessation programs, can decrease the incidence of cancers linked to these risk factors.
Primary prevention: Strategies like vaccines for HPV and HBV, reducing tobacco use, and regulating carcinogenic exposures could drive down incidence of certain cancers. Models show primary prevention could prevent 50% or more of cancer cases.
Secondary prevention: - Innovations in early detection through blood tests, AI imaging, nano-sensors and more personalized screening could allow earlier intervention, stopping progression to a malignant process for many patients. The potential impact of novel cancer preventive strategies on overall epidemiology is diverse and promising, but also complex and challenging to predict. Here's a breakdown of potential effects:
Reduced cancer incidence: Effective interventions could lead to a significant decrease in the number of new cancer cases across various types. This could be achieved through primary prevention - targeting modifiable risk factors like smoking, unhealthy diet, and obesity through lifestyle interventions, dietary modifications, and vaccination (e.g., HPV vaccine for cervical cancer). Implementing advanced screening technologies and personalized risk assessment models to identify precancerous lesions and early-stage cancers will allow timely intervention and significantly improved prognosis.
Chemoprevention: Utilizing medication or dietary supplements to prevent or delay the development of cancer in individuals at high risk. Widespread vaccination programs, like those for Human Papillomavirus (HPV) to prevent cervical and other cancers, can lead to a significant reduction in cancer incidence.
Early detection and intervention: Novel screening techniques, including liquid biopsies and advanced imaging technologies, can detect cancers at earlier stages when they are more treatable. Early intervention in pre-cancerous conditions can prevent the progression to full-blown cancer Mortality from cancer will decrease with a reduction in the number of advanced cancer cases through early detection and prevention. Improved survival rates will follow.
However, the epidemiologic benefit assumes equitable implementation across populations, overcoming barriers due to cost, health literacy, geography, tech access and trust. Thus, effective policy, regulation and health system advance are equally crucial to unlock the full potential of rapidly advancing prevention science. If cutting-edge primary, secondary and tertiary strategies can be made accessible globally, models estimate an over 50% reduction in mortality are realistically attainable. Improved survival rates will also be generated by novel approaches to metastatic cancer. Targeted therapies to specific mutations or molecular pathways unique to specific cancer types will lead to more effective and less toxic treatment options. Harnessing the immune system to recognize and attack cancer cells, already offers a promising approach for some cancers that previously had limited treatment options.
Healthcare resource allocation: Effective prevention strategies can reduce the burden on healthcare systems by decreasing the need for expensive, late-stage cancer treatments in some cancers, bending the growth curves sharply downward by 2050. But without thoughtful access promotion, advances may worsen disparities. Getting game-changing prevention to all populations should be the focus to alter cancer's future epidemiology. Population and socioeconomic background are crucial. Addressing health disparities and fostering trust in healthcare systems are the key to maximizing the impact. The long-term effectiveness and potential side effects of some novel strategies require careful monitoring and evaluation. Continuous research and development are essential to optimize and refine preventative interventions. Issues surrounding informed consent, genetic testing, and data privacy need careful consideration when implementing novel strategies. Balancing individual autonomy with public health benefits will continue to be an ongoing challenge.
Overall, while the potential for novel cancer preventive strategies to revolutionize epidemiology is significant, their successful implementation necessitates a multifaceted approach addressing social, economic, and ethical considerations alongside scientific advance. Resources can be reallocated to other areas of healthcare, including further research into prevention and early detection. The impact of novel preventive strategies may vary globally, with high-income countries seeing more immediate benefits due to better access to healthcare and technology. Efforts will be needed to ensure low-middle income countries also benefit from advances in cancer prevention. Governments and health organizations may shift focus to implementing policies that promote healthy lifestyles, environmental protection, and access to preventive healthcare services.
In summary, novel cancer preventive strategies hold the potential to substantially alter the landscape of cancer epidemiology, reducing both the incidence and severity of cancer, and improving overall survival rates.
- How will the life-years-lost to cancer change?
Estimating life years lost (LYL) due to cancer involves comparing the expected lifespan of individuals without cancer to those diagnosed with cancer. The LYL due to cancer can be a powerful measure of the disease's impact on mortality, reflecting not just the number of deaths but the age at which those deaths occurred and the potential years of life that are lost as a result. Detailed, up-to-date global statistics on life years lost to cancer are not readily available in a consolidated form, as they can vary significantly by region, type of cancer, age at diagnosis, and over time due to changes in cancer incidence, survival rates, and population demographics. However, I can discuss general trends and factors that could influence how LYL due to cancer might change in the future:
Cancer incidence and mortality rates: If the incidence of high mortality cancers increases, or if survival rates for major cancers do not improve, LYL could increase. Conversely, if prevention, early detection, and treatment continue to improve, leading to lower mortality rates, LYL could decrease.
Aging populations: Most cancers are more common in older people. Unfortunately, I do not have enough context or details to provide a specific projection on life years lost due to cancer. Some factors that would help make a projection would be current life expectancy and cancer mortality rates by country. Life years lost is typically calculated based on the difference between life expectancy and age at death from cancer. Having current baseline data is therefore important. Cancer incidence and survival rate trends. Understanding historical trends and forecasts for cancer cases and outcomes would inform a projection. Survival rates have been improving for many cancers with advances in screening and treatment.
Demographic shifts: Aging populations in many countries contribute to more cancer diagnoses. Population growth in developing countries also impacts cancer rates and hence life years lost.
Lifestyle and risk factors: Trends related to smoking, obesity, environmental exposures may increase or decrease certain cancer rates. So, a comprehensive projection would synthesize data and models around demographics, cancer statistics, risk factors, and health systems capacity. The number of life years lost due to cancer is a significant and multifaceted issue. Here's what I can share:
Current situation: Globally in 2019, an estimated 250 million disability-adjusted life years (DALYs) were lost due to cancer, with 96.9% attributed to life years lost. This translates to roughly 2.5 million years of life lost to cancer each year. The burden varies across regions. Low-middle income countries experience a higher proportion of the global cancer burden despite having lower incidence rates. There is of course considerable variation by cancer type. Lung cancer contributes the most LYL globally, followed by colon and breast cancer. Lung cancer is common, occurs in relatively young people and has poor outcomes even at early stages. However, cancers with lower incidence rates can have a significant impact, especially if they affect younger individuals.
Predicting the future: Predicting future trends in life years lost due to cancer is complex. Several factors play a role:
- Improved preventive measures: such as early detection and lifestyle changes could reduce cancer incidence and improve survival rates, leading to fewer LYL.
- Advances in treatment: Effective therapies and personalized medicine could extend older adults. As the global population ages, the number of cancer cases and potentially the LYL due to cancer could increase, even if the age -specific incidence rates of cancer remain stable or decline.
- Improvements in cancer treatment: Advances in personalized medicine, Immunotherapy, targeted therapies, and early detection have improved survival rates for many cancers. Continued progress in these areas could lead to further reductions in LYL by extending the lives of people with cancer.
- Disparities in access: to cancer prevention, screening, and treatment services, both within and between countries, can affect cancer survival rates and, consequently, LYL. Efforts to reduce these disparities could lead to significant reductions in LYL due to cancer.
- Population aging: As the global population ages, the overall number of cancer diagnoses and deaths is likely to increase, potentially offsetting gains from prevention and treatment.
- Socio-economic factors: Access to healthcare, lifestyle choices, and environmental factors can significantly impact cancer burden. Addressing these inequalities is crucial for reducing disparities in YLLs.
Therefore, the future of life years lost due to cancer is frankly very uncertain. It depends on numerous interconnected factors, with the potential for both positive and negative developments. lead to earlier detection of cancers at stages when they are more treatable, potentially reducing LYL. To forecast changes in LYL due to cancer, one would need to model these and other factors, taking into account regional differences, anticipated advancements in medical science, and potential shifts in health policies and public health initiatives. Such analyses would likely require complex statistical or computational models and could be subject to significant uncertainty, especially regarding future breakthroughs in treatment or changes in societal behaviour patterns.
Lower cancer incidence and improved survival rates could translate to a significant reduction in the overall healthcare burden related to cancer. This could include decreased cancer treatment costs, reduced pressure on medical resources, and improved quality of life for patients and their families. Challenges and considerations to ensure equitable access to novel preventive strategies across various intervention can shift cancer towards being a manageable chronic disease rather than a fatal one. There will be a shift in cancer types as certain types of cancers become less common due to effective prevention (e.g. lung cancer with reduced smoking rates), the relative prevalence of other cancer types may increase. The aging population might lead to an increase in cancers more common in older age groups, so actually reducing the LYL simply because of reduced natural life expectancy in the older population.
Purchase the full report to read the remaining chapters where we answer key questions regarding Diagnosis, Treatment, Economics, Workforce, and Service Configuration.